 To help and support our clients we are providing a limited number of 250 daily discount codes. Hurry, first come, first served!
To help and support our clients we are providing a limited number of 250 daily discount codes. Hurry, first come, first served!
ISO 13485 Training FAQs
ISO 13485 is an international standard for quality management systems in medical devices. It ensures organisations meet regulatory requirements, provide safe and effective products, enhance customer trust, and improve overall operational efficiency.
While primarily focused on medical devices, ISO 13485 applies to any organisation involved in designing, producing, or supplying healthcare-related products, ensuring quality and compliance with regulatory requirements in the medical field.
ISO 13485 is a standard for quality management systems, whereas GMP (Good Manufacturing Practice) focuses on manufacturing processes. ISO 13485 ensures compliance, while GMP emphasises consistent product quality and safety.
The course is designed to be accessible to all levels, making it suitable for both beginners and experienced professionals. It covers foundational concepts while offering in-depth insights into personal and organisational growth strategies.
ISO 13485 Training ensures improved compliance with global regulations, enhances organisational efficiency, reduces risks, and boosts professional credentials for individuals working within the medical device and healthcare industry.
ISO 13485 Certification aims to establish robust quality management systems, ensuring organisations deliver safe, high-quality medical devices that comply with international regulations and build stakeholder trust.
Quality managers, Regulatory Professionals, Auditors, and Personnel involved in designing, manufacturing, or distributing medical devices benefit greatly from ISO 13485 Medical Devices Training.
The prerequisites for this course are based on the course specifications and the target group of professionals it serves. Check the respective course page of the course that you are planning to take to know about its prerequisites.
Holding ISO 13485 Certification demonstrates expertise in medical device quality management, enhances professional credibility, improves career opportunities, and ensures compliance with international regulatory standards.
Key steps include understanding requirements, conducting a gap analysis, developing a quality management system, implementing procedures, training employees, and undergoing an audit to achieve certification.
In this training course, delegates will have intensive training with our experienced instructors, a digital delegate pack consisting of important notes related to this course, and a certificate after course completion.
These courses can take upto 1-5 days during which delegates participate in intensive learning sessions that cover various course topics.
Yes, The Knowledge Academy offers 24/7 support via phone & email before attending, during, and after the course. Our customer support team is available to assist and promptly resolve any issues you may encounter.
The Knowledge Academy provides flexible self-paced training for this course. Self-paced training is beneficial for individuals who have an independent learning style and wish to study at their own pace and convenience.
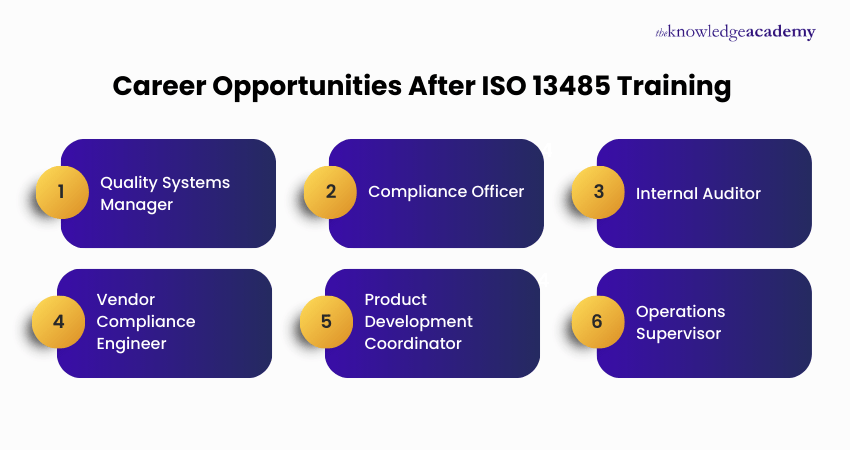
Roles include Quality Manager, Regulatory Affairs Specialist, Quality Auditor, Compliance Officer, and Medical Device Consultant, offering opportunities in healthcare manufacturing, regulatory agencies, and consulting firms.
Yes, we provide corporate training for this course, tailored to fit your organisation’s requirements.
Delegates learn ISO 13485 principles, quality system implementation, risk management, regulatory compliance, internal auditing, and techniques to maintain effective quality management in medical device production.
Professionals involved in medical device quality management, regulatory compliance, or production, as well as those preparing for ISO 13485 audits, should attend these certification courses.
If you are unable to access your training, contact the support team at The Knowledge Academy via their customer service email or phone number provided on their website for prompt assistance and resolution of your issue.
ISO 13485 Training enhances knowledge of medical device quality management, ensures compliance with global regulations, improves career opportunities, and provides the skills needed to implement and maintain effective quality systems within medical device organisations.
No formal qualification is required; however, prior knowledge of quality management systems or experience in medical device industries is beneficial for understanding ISO 13485.
After completing the training, professionals can implement ISO 13485 systems, prepare for certification audits, enhance career opportunities, and apply their knowledge to ensure regulatory compliance in medical device organisations.
Yes, most ISO 13485 Training Courses include an exam to assess the delegate's understanding of quality management principles and regulatory compliance. Passing the exam typically results in receiving a certification.
Please see our ISO 13485 Training available in Philippines
The Knowledge Academy is one of the Leading global training provider for ISO 13485 Training.
The training fees for ISO 13485 Training in Philippines starts from $3195

Why we're the go to training provider for you

Best price in the industry
You won't find better value in the marketplace. If you do find a lower price, we will beat it.

Trusted & Approved
Recognised by leading certification bodies, we deliver training you can trust.

Many delivery methods
Flexible delivery methods are available depending on your learning style.

High quality resources
Resources are included for a comprehensive learning experience.




"Really good course and well organised. Trainer was great with a sense of humour - his experience allowed a free flowing course, structured to help you gain as much information & relevant experience whilst helping prepare you for the exam"
Joshua Davies, Thames Water



Looking for more information on ISO 13485 Training
 Top 20 ISO 13485 Audit Questions and Answers
Top 20 ISO 13485 Audit Questions and Answers ISO 13485 Auditor Salary: A Comprehensive Guide
ISO 13485 Auditor Salary: A Comprehensive Guide ISO 13485 Auditor Jobs: Roles, Roadmap, and Salary
ISO 13485 Auditor Jobs: Roles, Roadmap, and Salary ISO 13485 Requirements: Everything You Need to Know
ISO 13485 Requirements: Everything You Need to Know Difference Between ISO 9001 and ISO 13485: A Complete Guide
Difference Between ISO 9001 and ISO 13485: A Complete Guide ISO 13485 Compliance: Requirements and Benefits
ISO 13485 Compliance: Requirements and Benefits Benefits of ISO | Business Advantages
Benefits of ISO | Business Advantages Why is ISO 13485 Important for Medical Device Quality?
Why is ISO 13485 Important for Medical Device Quality? Examples and Explanation of the ISO 13485 Principles
Examples and Explanation of the ISO 13485 Principles 













 If you wish to make any changes to your course, please
If you wish to make any changes to your course, please


