We may not have the course you’re looking for. If you enquire or give us a call on +44 1344 203999 and speak to our training experts, we may still be able to help with your training requirements.
We ensure quality, budget-alignment, and timely delivery by our expert instructors.

A small error in a medical device can have life-altering consequences. So, how do leading companies ensure their products meet the highest safety and quality standards? It starts with understanding What is ISO 13485. In case, you are designing surgical tools or producing diagnostic kits, every detail matters; and ISO 13485 provides the trusted framework to get it right.
But What is ISO 13485 about? It’s more than compliance; it’s a global commitment to patient safety, continuous improvement, and operational excellence. Let’s dive into the world where quality isn’t just expected; it’s engineered.
Table of Contents
1) What is ISO 13485?
2) Latest Standards of ISO 13485
3) ISO 13485 Documentation Requirements
4) The Eight Sections of ISO 13485
5) What is the Difference Between ISO 13485 and EN ISO 13485?
6) ISO 13485:2016 vs 13485:2012
7) What is the Difference Between ISO 9001 and ISO 13485?
8) Importance of ISO 13485
9) Conclusion
What is ISO 13485?
ISO 13485 is an internationally recognised Quality Management System (QMS) standard for the medical device industry. It ensures companies meet regulatory and customer requirements by focusing on product safety, risk management, and consistent quality. The standard applies to organisations of all sizes involved in designing, producing, or servicing medical devices and related services.
Latest Standards of ISO 13485
After learning What is ISO 13485, it is time to learn about the current developments in the standard. The latest ISO 13485 standard is ISO 13485:2016, last reviewed in 2020. This revision brings several significant changes and improvements to the standard. Understanding the Benefits of ISO 13485 in this updated version is crucial, as it highlights how the changes can further enhance product quality, regulatory compliance, and operational efficiency in the medical device industry. The major changes made in the latest version are as follows:
a) The latest version emphasises effective document control. It requires organisations to establish and maintain documented procedures for document management, including revision control, distribution, and access.
b) ISO 13485:2016 places greater emphasis on risk management throughout the product lifecycle. It requires organisations to identify, assess, and mitigate risks associated with their devices, ensuring patient safety.
c) The revised standard adopts a process-based approach, emphasising the importance of clearly defined procedures, documentation, and traceability. This enables organisations to streamline their operations and improve overall efficiency.
d) ISO 13485:2016 considers the entire lifecycle of a Medical Device, including its design, development, production, installation, and post-market activities. This comprehensive approach helps organisations address quality requirements at each stage, ensuring consistent product performance.
e) It places greater emphasis on Supply Chain Management. It requires organisations to establish processes to ensure that quality components and materials are used in their devices.
f) The revised standard also promotes risk-based decision-making, encouraging organisations to use risk assessments to inform their Quality Management processes and decision-making activities.
Enhance your career with ISO 13485 Training Courses – Start mastering quality management today!
ISO 13485 Documentation Requirements
ISO 13485 requires the organisation to document various aspects of its QMS and its Medical Devices. Let's explore the documentation Requirements of ISO 13485:
1) Quality Manual
The quality manual is an important document that defines the scope, structure, and main elements of the QMS. It includes the quality policy, the quality objectives, and the references to the relevant procedures and records. The quality manual also defines the roles undertaken by the organisation under applicable regulatory requirements.
2) Medical Device File
The Medical Device file is a document that contains the description, specifications, and records of the Medical Devices. It includes the following:
1) Intended use
2) The indications for use
3) The labelling
4) The instructions for use
5) The manufacturing instructions
6) Inspection instructions
7) Packaging instructions
8) Storage instructions
9) Handling instructions
10) Distribution specifications and procedures
11) Monitoring specifications and procedures
The Medical Device file also provides evidence of the conformity and traceability of the Medical Devices.
3) Document Control
Document control is a procedure that ensures that the documents required by the QMS and the Medical Device are approved, updated, identified, available, legible, and retrievable. Document control also prevents the unintended use of obsolete documents and applies to both internal and external documents.
4) Control of Records
Control of records is a procedure that ensures that the records of the QMS and Medical Devices are identified, collected, indexed, stored, protected, accessed, retained, and disposed of in an organised manner. Records provide evidence of the implementation and effectiveness of the QMS and the Medical Device, as well as the compliance with the regulatory requirements
Learn how to understand the QMS requirements for Medical Devices with our ISO 13485 Foundation Training – join today!
The Eight Sections of ISO 13485
ISO 13485 outlines the requirements for a Quality Management System (QMS) to ensure the consistent production of medical devices and related services that meet customer needs and regulatory standards. The standard applies to organisations of all sizes and types, except where explicitly stated. It also covers both medical devices and associated services provided by the organization.
The standard is structured into eight key sections:
1) Scope: Defines the purpose, applicability, and limitations of ISO 13485.
2) Normative References: Lists essential documents referenced within the standard.
3) Terms and Definitions: Provides key terminology to ensure consistency in understanding.
4) Quality Management System: Establishes the general QMS requirements, including documentation.
5) Management Responsibility: Outlines leadership duties in maintaining and improving the QMS.
6) Resource Management: Covers the allocation of human, infrastructure, and work environment resources.
7) Product Realisation: Details the process from design and development to production and delivery.
8) Measurement, Analysis, and Improvement: Focuses on monitoring, auditing, and continuous improvement to maintain compliance.
Master implementation with ISO 13485 Lead Implementer Certification. Learn to apply the standard and drive quality improvements.
What is the Difference Between ISO 13485 and EN ISO 13485?
ISO 13485 is a globally recognised standard developed by the International Organization for Standardization (ISO) to establish a Quality Management System (QMS) for companies in the medical device industry. It outlines the key requirements businesses must follow to ensure their medical devices consistently meet regulatory and customer expectations.
EN ISO 13485, on the other hand, is the European version of this standard, tailored for use within the EU. While the core requirements of both versions are identical, EN ISO 13485 includes additional reference tables that link the standard to key EU medical device directives (90/385/EEC, 93/42/EEC, and 98/79/EC). These tables help companies align with European regulations more easily.
With the introduction of the EU Medical Device Regulation (EU MDR) in 2017, which replaced these directives, a revised version of EN ISO 13485 is expected to better align with the new regulatory framework.

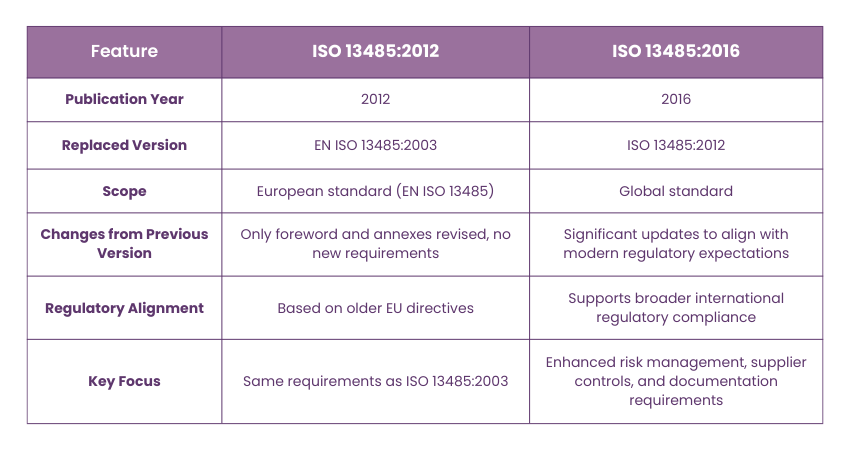
ISO 13485:2016 vs 13485:2012
The European standard EN ISO 13485:2012; titled Medical Devices - Quality Management Systems - Requirements for Regulatory Purposes,was officially approved by CEN on January 24, 2012. This version replaced EN ISO 13485:2003, but with minimal changes.
While the global ISO 13485:2003 standard itself remained unchanged, the European edition (EN ISO 13485:2012) saw updates only in the foreword and annexes. This means there were no new or modified requirements between ISO 13485:2012 and ISO 13485:2003, making the transition seamless for businesses already compliant with the 2003 version.
What is the Difference Between ISO 9001 and ISO 13485?
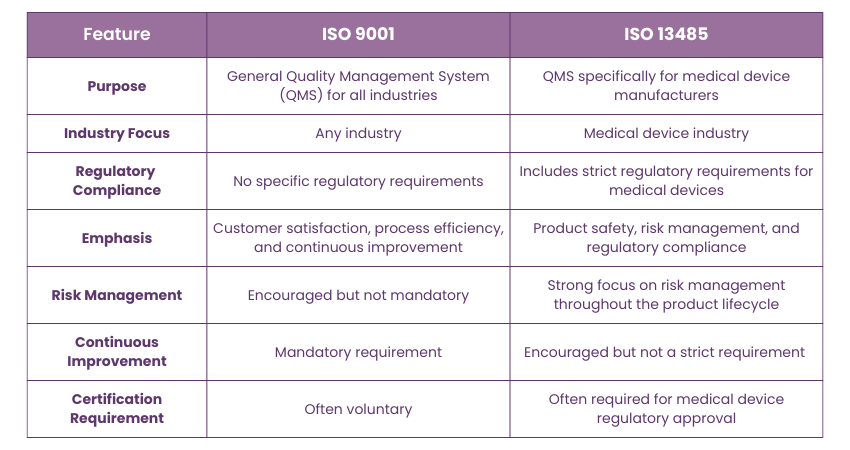
Although ISO 13485:2016 is an independent standard, it is built upon the foundation of ISO 9001:2008. The key difference lies in their scope:
1) ISO 9001 is a general Quality Management System (QMS) standard, applicable to any organization across any industry.
2) ISO 13485, while based on ISO 9001, adds specific requirements for companies involved in medical device manufacturing to ensure compliance with regulatory and safety standards.
Essentially, ISO 9001 focuses on customer satisfaction and continuous improvement, while ISO 13485 emphasizes risk management, regulatory compliance, and product safety in the medical device sector.
Importance of ISO 13485
ISO 13485 holds significant importance in the Medical Device industry. Let’s explore the Importance of ISO 13485 certification for organisations:
1) Regulatory Compliance
ISO 13485 aligns with regulatory requirements in various regions, such as the European Union's Medical Device Regulation (MDR) and the U.S. Food and Drug Administration's Quality System Regulation (QSR). Compliance with this standard demonstrates compliance with these regulations, facilitating market access for Medical Devices.
Become an expert with ISO 13485 Lead Auditor Training. Gain the skills to conduct audits and ensure compliance effectively.
2) Enhanced Product Quality
ISO 13485 promotes implementing robust Quality Management Systems, focusing on risk management, design and development processes, production controls, and post-market surveillance. By adhering to the standard, organisations can identify and mitigate potential risks, resulting in safer and more reliable Medical Devices.
3) Customer Confidence
Adherence to this ISO standard shows a manufacturer's commitment to delivering high-quality products that meet customer expectations. It enhances customer confidence by demonstrating compliance with internationally recognised standards and a dedication to continuous improvement. This, in turn,his can lead to improved customer trust and satisfaction.
4) Streamlined Processes
ISO 13485 emphasises process-based approaches, documentation, and traceability. By implementing efficient processes, organisations can enhance productivity, reduce errors, and minimise costs. A well-structured Quality Management System helps streamline operations and ensures consistent adherence to regulatory and customer requirements.
5) International Recognition
It is globally recognised and accepted, providing a common language for organisations operating in the Medical Device industry. This recognition allows manufacturers to expand their market reach, attract international customers, and collaborate with partners worldwide. ISO 13485 Certification shows an organisation's commitment to meeting international quality standards.
6) Risk Management
ISO 13485 Auditor Salary often reflects the expertise required for effective risk management throughout the product lifecycle.Organisations can ensure patient safety and minimise adverse events by implementing a systematic risk assessment and mitigation approach. This proactive approach to risk management enables informed decision-making and the implementation of appropriate measures to mitigate potential hazards.
Looking to ace your ISO 13485 audit? Get expert insights with our ISO 13485 Audit Questions and Answers. Start preparing today!
Conclusion
To conclude, understanding What is ISO 13485 empowers organisations to deliver medical devices that meet global safety and quality expectations. It’s a mark of trust, professionalism, and commitment to patient wellbeing. By embedding these standards into your processes, you not only achieve compliance but also drive innovation and strengthen your place in a competitive market.
Learn internal auditing with our ISO 13485 Internal Auditor Training – sign up now!
Frequently Asked Questions
Who Certifies ISO 13485?

ISO 13485 certification is issued by accredited certification bodies, not by ISO itself. These bodies assess a company's Quality Management System (QMS) for compliance with ISO 13485 standards. To ensure legitimacy, choose a certification body accredited by recognized organisations like UKAS, ANAB, or other national accreditation bodies.
How Do I Know if My Company is ISO 13485 Certified?

To verify ISO 13485 certification, check your company's certification documents issued by an accredited body. You can also confirm certification status by searching the certification body's official database or requesting a copy of the valid ISO 13485 certificate from your quality assurance team.
What are the Other Resources and Offers Provided by The Knowledge Academy?

The Knowledge Academy takes global learning to new heights, offering over 3,000 online courses across 490+ locations in 190+ countries. This expansive reach ensures accessibility and convenience for learners worldwide.
Alongside our diverse Online Course Catalogue, encompassing 19 major categories, we go the extra mile by providing a plethora of free educational Online Resources like News updates, Blogs, videos, webinars, and interview questions. Tailoring learning experiences further, professionals can maximise value with customisable Course Bundles of TKA.
What is The Knowledge Pass, and How Does it Work?

The Knowledge Academy’s Knowledge Pass, a prepaid voucher, adds another layer of flexibility, allowing course bookings over a 12-month period. Join us on a journey where education knows no bounds.
What are the Related Courses and Blogs Provided by The Knowledge Academy?

The Knowledge Academy offers various ISO 13485 Training, including ISO 13485 Foundation, ISO 13485 Lead Auditor and many more. These courses cater to different skill levels, providing comprehensive insights into What is ISO 13485.
Our Health and Safety Blogs cover a range of topics related to ISO 13485, offering valuable resources, best practices, and industry insights. Whether you are a beginner or looking to advance your Compliance skills, The Knowledge Academy's diverse courses and informative blogs have you covered.

The Knowledge Academy is a world-leading provider of professional training courses, offering globally recognised qualifications across a wide range of subjects. With expert trainers, up-to-date course material, and flexible learning options, we aim to empower professionals and organisations to achieve their goals through continuous learning.

Upcoming Health & Safety Resources Batches & Dates
Date
 ISO 13485 Foundation
ISO 13485 Foundation
Mon 17th Aug 2026
Mon 16th Nov 2026
Mon 7th Dec 2026





 Top Rated Course
Top Rated Course








 If you wish to make any changes to your course, please
If you wish to make any changes to your course, please


